  
It is a physical phenomenon explained by Ernest Rutherford in 1911 that led to the development of the planetary Rutherford model of the atom and eventually the Bohr model. Can you explain this answer? tests, examples and also practice Class 9 tests.Rutherford scattering is the elastic scattering of charged particles by the Coulomb interaction. Can you explain this answer? theory, EduRev gives you anĪmple number of questions to practice The conclusion of Rutherford’s scattering experiment does not includea)a-particles can come within the distance of order of 10-14m of the nucleus.b)The radius of nucleus is less than 10-14m.c)Scattering follows coulomb’s law, i.e., same charges repel each other.d)The positively charged particles of atom move with very high velocit圜orrect answer is option 'D'. Can you explain this answer? has been provided alongside types of The conclusion of Rutherford’s scattering experiment does not includea)a-particles can come within the distance of order of 10-14m of the nucleus.b)The radius of nucleus is less than 10-14m.c)Scattering follows coulomb’s law, i.e., same charges repel each other.d)The positively charged particles of atom move with very high velocit圜orrect answer is option 'D'. Can you explain this answer?, a detailed solution for The conclusion of Rutherford’s scattering experiment does not includea)a-particles can come within the distance of order of 10-14m of the nucleus.b)The radius of nucleus is less than 10-14m.c)Scattering follows coulomb’s law, i.e., same charges repel each other.d)The positively charged particles of atom move with very high velocit圜orrect answer is option 'D'. The conclusion of Rutherford’s scattering experiment does not includea)a-particles can come within the distance of order of 10-14m of the nucleus.b)The radius of nucleus is less than 10-14m.c)Scattering follows coulomb’s law, i.e., same charges repel each other.d)The positively charged particles of atom move with very high velocit圜orrect answer is option 'D'. Can you explain this answer? defined & explained in the simplest way possible. Here you can find the meaning of The conclusion of Rutherford’s scattering experiment does not includea)a-particles can come within the distance of order of 10-14m of the nucleus.b)The radius of nucleus is less than 10-14m.c)Scattering follows coulomb’s law, i.e., same charges repel each other.d)The positively charged particles of atom move with very high velocit圜orrect answer is option 'D'. Can you explain this answer? covers all topics & solutions for ClExam.įind important definitions, questions, meanings, examples, exercises and tests below for The conclusion of Rutherford’s scattering experiment does not includea)a-particles can come within the distance of order of 10-14m of the nucleus.b)The radius of nucleus is less than 10-14m.c)Scattering follows coulomb’s law, i.e., same charges repel each other.d)The positively charged particles of atom move with very high velocit圜orrect answer is option 'D'. 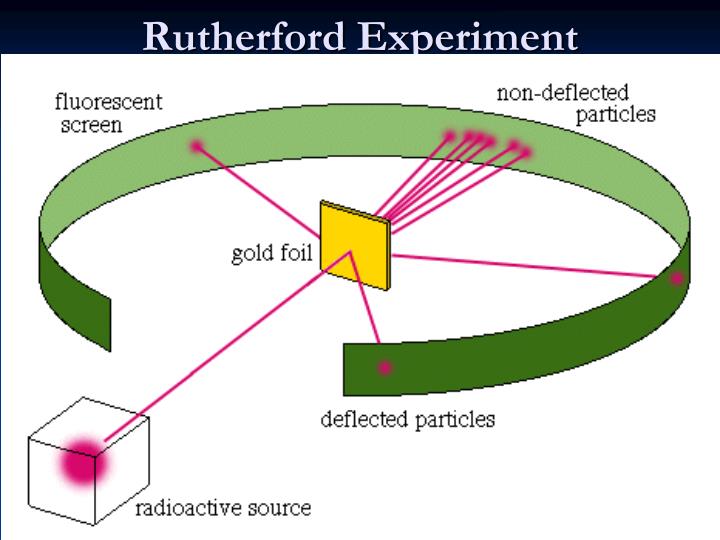
Information about The conclusion of Rutherford’s scattering experiment does not includea)a-particles can come within the distance of order of 10-14m of the nucleus.b)The radius of nucleus is less than 10-14m.c)Scattering follows coulomb’s law, i.e., same charges repel each other.d)The positively charged particles of atom move with very high velocit圜orrect answer is option 'D'. The Question and answers have been prepared Can you explain this answer? for Clis part of Class 9 preparation. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
